Interfacial asymmetry of water published in J. Chem. Phys.
A. Shandilya, K. Schwarz and R. Sundararaman, “Interfacial water asymmetry at ideal electrochemical interfaces”, J. Chem. Phys. 156, 014705 (2022)
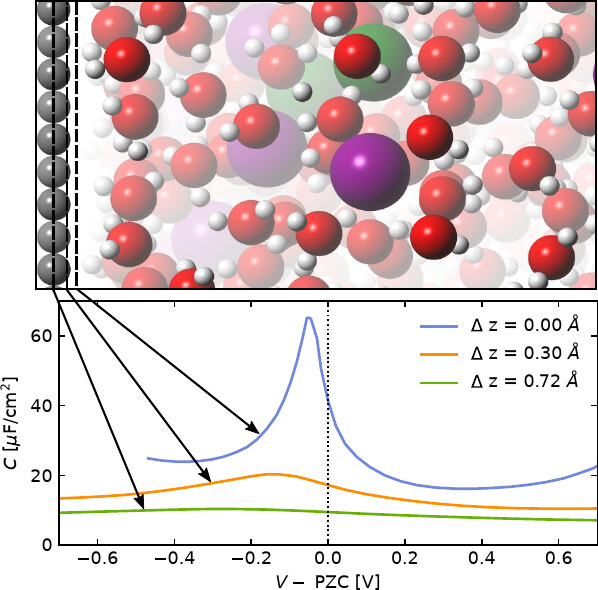
Controlling electrochemical reactivity requires a detailed understanding of the charging behavior and thermodynamics of the electrochemical interface. Experiments can independently probe the overall charge response of the electrochemical double layer by capacitance measurements and the thermodynamics of the inner layer with potential of maximum entropy measurements. Relating these properties by computational modeling of the electrochemical interface has so far been challenging due to the low accuracy of classical molecular dynamics (MD) for capacitance and the limited time and length scales of ab initio MD.
Here, we combine large ensembles of long-time-scale classical MD simulations with charge response from electronic density functional theory to predict the potential-dependent capacitance of a family of ideal aqueous electrochemical interfaces with different peak capacitances. We show that while the potential of maximum capacitance varies, this entire family exhibits an electrode charge of maximum capacitance (CMC) between −2.9 and −2.2 μC/cm2, regardless of the details in the electronic response. Simulated heating of the same interfaces reveals that the entropy peaks at a charge of maximum entropy (CME) of −5.1 ± 0.6 μC/cm2, in agreement with experimental findings for metallic electrodes. The CME and CMC both indicate asymmetric response of interfacial water that is stronger for negatively charged electrodes, while the difference between CME and CMC illustrates the richness in behavior of even the ideal electrochemical interface.
